|
11/19/2022 0 Comments Nuclear fission uranium bomb 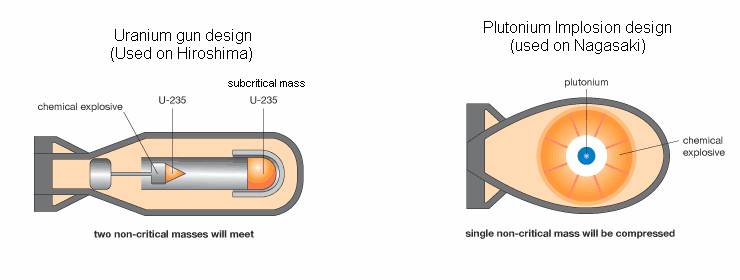
Glenn Seaborg, an American chemist and physicist, received the Nobel Prize in physics in 1951 for discovery of several transuranic elements, including plutonium. It not only proved that the chain reaction was possible, it began the era of nuclear reactors. This first “atomic pile”, built in a squash court at the University of Chicago, used carbon blocks to thermalize neutrons. The first major step was made by Enrico Fermi and his group in December 1942, when they achieved the first self-sustained nuclear reactor. Robert Oppenheimer (1904–1967), whose talent and ambitions made him ideal, was chosen to head the project. It was carried out in remote locations, such as Los Alamos, New Mexico, whenever possible, and eventually came to cost billions of dollars and employ the efforts of more than 100,000 people. The top secret Manhattan Project was a crash program aimed at beating the Germans. It was not until December 6, 1941, the day before the Japanese attack on Pearl Harbor, that the United States made a massive commitment to building a nuclear bomb. It was sent in August of 1939, just before the German invasion of Poland that marked the start of World War II. The letter was for President Franklin Roosevelt, warning of the German potential to build extremely powerful bombs of a new type. 
Leo Szilard, an escaped Hungarian physicist, took a draft of a letter to Einstein, who, although pacifistic, signed the final version. It was felt that his help was needed to get the American government to make a serious effort at nuclear weapons as a matter of survival. None was more famous or revered than Einstein. It seemed that the military value of uranium had been recognized in Nazi Germany, and that a serious effort to build a nuclear bomb had begun.Īlarmed scientists, many of them who fled Nazi Germany, decided to take action. Within months after the announcement of the discovery of fission, Adolf Hitler banned the export of uranium from newly occupied Czechoslovakia. The enormous energy known to be in nuclei, but considered inaccessible, now seemed to be available on a large scale. The possibility of a self-sustained chain reaction was immediately recognized by leading scientists the world over. Fermi, among others, soon found that not only did neutrons induce fission more neutrons were produced during fission. The discovery of fission, made by two German physicists, Otto Hahn and Fritz Strassman, was quickly verified by two Jewish refugees from Nazi Germany, Lise Meitner and her nephew Otto Frisch. The world was in turmoil when fission was discovered in 1938. Explain the ill effects of nuclear explosion. 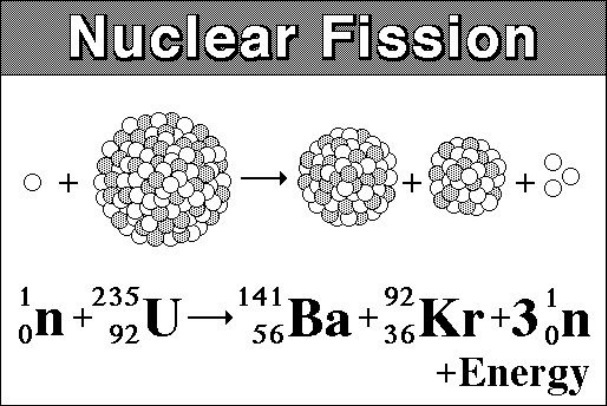
Discuss different types of fission and thermonuclear bombs.This filtering process is called enrichment and Professor Poliakoff from periodicvideos.By the end of this section, you will be able to: Only the U-235 isotope is fissile (able to undergo fission) and mined uranium must be processed to remove the U-238 isotope. Without that control, fission occurs rapidly and all of the energy is released in a very short period, resulting in a nuclear explosion. If the fission process is controlled, by limiting the number of available neutrons, then it can be used in a nuclear power station. These neutrons can go on to be absorbed by other U-235 atoms, causing further fission. Notice how the fission process shown above produces 3 new neutrons in addition to the fission fragments. We can calculate how much energy is released using Einstein’s famous equation For each U-235 atom that undergoes fission, a large quantity of energy is released. The “missing mass” is converted into energy. The total mass of the original U-235 atom and the single neutron is greater than the combined mass of the two fission fragments and the three new neutrons. The animation below is a representation of the nucleus of a U-235 atom absorbing a passing (blue) neutron and splitting into two fission fragments as a result.Īnimated gif showing fission of U-235 by Stephan-Xp Atoms of U-235 have a special property, they can absorb low energy neutrons and then split up into two smaller atoms. U-235 (92 protons, 143 neutrons) 0.71% of uranium is this isotopeĪlthough less than 1% of all the uranium we can dig out of the ground is the U-235 isoptope, this is the one that gets most attention.U-238 (92 protons, 146 neutrons) 99.28% of uranium is this isotope.Uranium has several isotopes, all of which are naturally radioactive. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
